Livonia, Michigan, USA and Tel Aviv, Israel – Hygieia, a digital health company for insulin therapy announces a new partnership with TempraMed, a manufacturer of advanced temperature-based insulin storage solutions. This relationship brings the innovative technologies of both companies to a much broader audience, helping more people simplify and effectively manage their diabetes care.
Hygieia’s d-Nav® technology is the first FDA-cleared AI-driven system that autonomously adjusts insulin doses based on the unique needs of its user. This enables more frequent and optimal dose adjustments to better manage glucose levels. In addition to the app which handles dosing, d-Nav users are supported by a team of expert clinicians who review patient data and stay in frequent communication. The program has demonstrated compelling results, with studies showing nearly 90 percent of patients improve their A1c, in as early as three months.
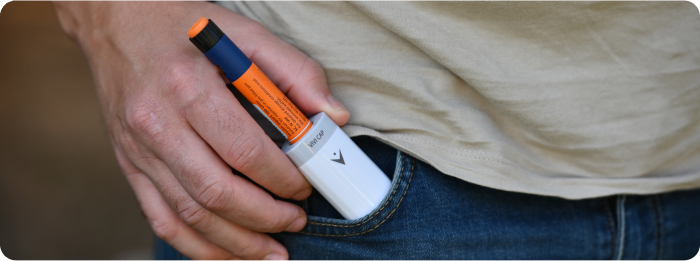
TempraMed’s flagship product, the VIVI Cap, is a lightweight, reusable insulin pen cap designed for those who use insulin and/or weight loss medications. The VIVI Cap ensures medication efficacy by maintaining optimal temperatures. Available globally through online platforms including the TempraMed website, Amazon, Walmart, Walgreens, and various distributors, the VIVI Cap ensures safe and convenient storage of diabetes medication.
“Our partnership with TempraMed allows us to continue to deliver value to our patients and increases our outreach to prospective patients,” said Eran Bashan, CEO of Hygieia. “It is important for people who use insulin to have solutions and products that help them manage their diabetes and live better lives. There is much to discover in this relationship, and I am excited to see what the future holds.”
About TempraMed
TempraMed is at the forefront of creating and delivering groundbreaking personal storage solutions for injectable medications. Our mission is to address the long-standing gap in the market for individuals who use injectable medications and seek to embrace a lifestyle marked by greater spontaneity, convenience, and safety. TempraMed develops patented sustainable energy products that provide ‘Last Mile’ control and management to all injectable and temperature sensitive pharmaceuticals. TempraMed’s first launched product VIVI Cap insulin temperature shield pen cap, is scientifically validated, has FDA, CE, and other regulatory registrations, and is used by tens of thousands worldwide. VIVI Cap provides 24/7/365 years of temperature protection for insulin without the need for any user intervention.
About Hygieia
Hygieia is the developer of d-Nav®, the first FDA-cleared technology to autonomously adjust insulin doses directly to the patient. d-Nav’s AI-powered technology automatically interprets data and independently initiates insulin management. The d-Nav Insulin Management Program adds clinical support that helps people with type 2 diabetes adjust their insulin doses according to their body’s changing insulin needs. Studies have shown that nearly 90 percent of people using d-Nav report lower A1c in just three months* without increasing the risk for hypoglycemia**. The d-Nav Insulin Management Program has been peer-reviewed with studies published in leading medical journals. Hygieia is headquartered in Livonia, Michigan. Learn more at d-Nav.com.
* John E Schneider et al. Impact of a Novel Insulin Management Service on Non-insulin Pharmaceutical Expenses. J Health Econ Outcomes Res. 2018 Feb 20;6(1):53-62. https://jheor.org/article/9783
** Richard M Bergenstal et al. Automated insulin dosing guidance to optimize insulin management in patients with type 2 diabetes: a multicenter, randomized controlled trial. Lancet 2019 Mar 16;393(10176):1138-1148. https://pubmed.ncbi.nlm.nih.gov/30808512/
d-Nav is prescribed by medical professionals and is accessible via Hygieia’s clinic in Michigan or through their endocrinologist clinical partners in select states.
For more information about Hygieia, please visit https://d-nav.com/. For more information about VIVI Cap, please visit www.tempramed.com.
Media Contact:
Theresa Gdowski, theresa.gdowski@hygieia.com